The CNS places the electronic file corresponding to the description of the authorisation file's layout (.xsd) at the disposal of the laboratory. Mandatory areas and data are described in the specifications under Appendix X. The formatting and structures published on the CNS website must be used. Any technical changes to the layout are notified to the laboratory that is given a period of 3 months to comply.
Within the framework of the materialisation of the authorisation request, the request must be filled out in accordance with the template provided for in appendix IX of the specifications. This document is published on the CNS website.
Processing of authorisation requests
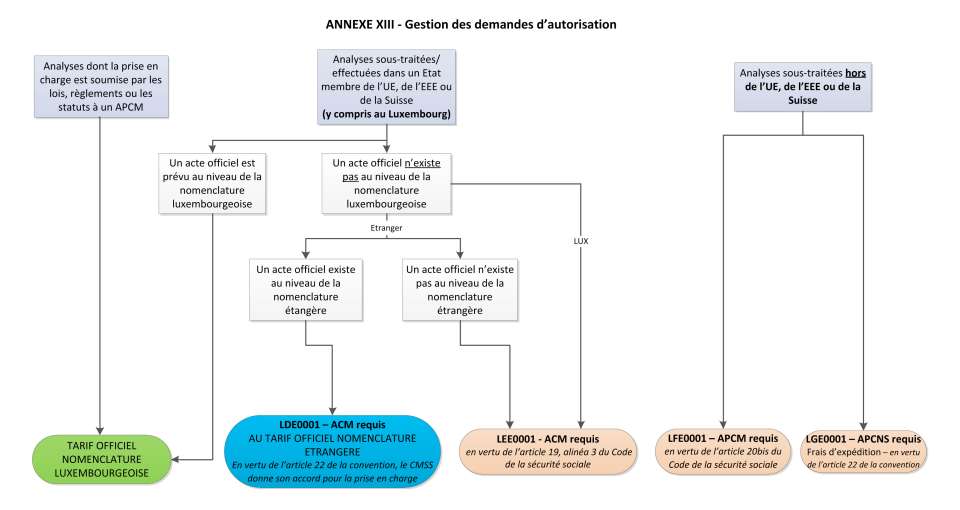
In accordance with provisions of article 23 of the specifications relating to the standard documents and forms agreed upon between the Luxembourg Federation for medical analyses laboratories and the CNS, the following collective codes are used depending on the authorisation procedure applied:
LDE0001 - ACM required
Analysis outsourced/performed in a Member State of the EU, the EEA or Switzerland, for which an official code is not provided for in the Luxembourg nomenclature of acts delivered by medical analyses laboratories, but for which an official act exists in the nomenclature of analyses covered by the legal health insurance scheme of the country (EU, EEA or Switzerland) where the analysis is carried out. In this case, the procedure as dictated by article 22 of the agreement is applicable. Paragraph 2 states:
Lorsque l’analyse a été transmise et effectuée dans un laboratoire situé en dehors du Grand-Duché de Luxembourg, mais dans un pays de l’UE, de l’EEE ou en Suisse et qu’un tarif y relatif n’est pas prévu par la nomenclature des actes et services des laboratoires d’analyses médicales et de biologie clinique au Luxembourg, elle est facturée par le laboratoire transmetteur conformément au tarif officiel opposable à l’assurance maladie légale dans l’Etat dans lequel l’analyse a été effectuée, à condition qu’elle tombe dans le champ d’application matériel de l’assurance maladie au Luxembourg, et que le Contrôle médical de la sécurité sociale ait donné son accord pour la prise en charge.
LEE0001 - ACM required
Exceptional analysis not found in either the Luxembourg nomenclature of acts provided by medical analyses laboratories or the nomenclature of analyses covered by the legal health insurance scheme of the country (EU, EEA or Switzerland) where it is carried out. The procedure as stated in article 19, paragraph 3 of the Social Security Code is applicable.
LFE0001 - APCM required
Exceptional analysis, outsourced outside of the EU, the EEA and Switzerland, not included in the nomenclature of acts provided by medical analyses laboratories. The procedure pursuant to article 20bis of the Social Security Code is applicable.
In accordance with article 19, paragraph 3 and article 20bis of the Social Security Code, the article 22 paragraph 3 of the agreement between the Luxembourg Federation for medical analyses laboratories and the CNS states:
A défaut d’un tarif officiel opposable à l’assurance maladie légale dans l’Etat membre dans lequel le laboratoire de collaboration est établi, mais que l’analyse tombe dans le champ d’application matériel de l’assurance maladie au Luxembourg, sans pour autant figurer dans la nomenclature des actes et services des laboratoires d’analyses médicales et de biologie clinique au Luxembourg, le remboursement par l’assurance maladie se fait sur base d’un tarif fixé par le Contrôle médical de la sécurité sociale assimilant la prestation exceptionnelle à une prestation de même importance. Il en est de même pour les analyses effectuées conformément aux conditions et modalités de la présente convention en dehors d’un pays de l’UE, de l’EEE ou de la Suisse.
LGE0001 - APCNS required
Shipping charges in case of an analysis transfer outside of the EU, the EEA or Switzerland. The procedure in accordance with article 22 of the agreement is applicable. Paragraph 5 states:
Dans les cas exceptionnels pour des analyses ne figurant pas dans la nomenclature des actes et services des laboratoires d’analyses de biologie médicale et ne pouvant pas être effectuées au Luxembourg ou dans un pays de l’UE, de l’EEE ou en Suisse, il est tenu compte des frais d’expédition exposés par le laboratoire transmetteur pour le transfert des prélèvements. La prise en charge des frais d’expédition est subordonnée à une autorisation préalable de la Caisse nationale de santé pour la transmission des analyses dans un pays en dehors de l’UE, de l’EEE et de la Suisse conformément à l’article 9 de la présente convention et à une justification des frais d’expédition par le laboratoire transmetteur.
Each analysis is subject to a separate request.
Requests for coverage of shipping costs outside of the EU, the EEA or Switzerland are made on the same request used for the analysis itself. Shipping costs are documented on a quote issued by the carrier. Shipping charges are covered only if the corresponding analyses are also covered.

